

众所众知,欧盟是由多个国家组成,查询全面的上市药品信息,也相对麻烦。除了欧洲药品管理局EMA,以外一些特定国家药品上市信息需要单独查询,就让我们盘点下吧:
这里有一份欧盟 (EU) 和欧洲经济区 (EEA) 不同成员国的国家药品注册清单。其中包含有关在这些国家/地区授权的药物的信息,包括指向医疗保健专业人员 (SmPC) 的产品信息和包装说明书的链接。这补充了 EMA 官网上发布的药物信息,该网站仅包括 EMA 评估的药物。有关欧盟不同药品授权途径的更多信息,请参阅药品授权。
- 第一个就是EMA授权的药品:
https://www.ema.europa.eu/en/medicines
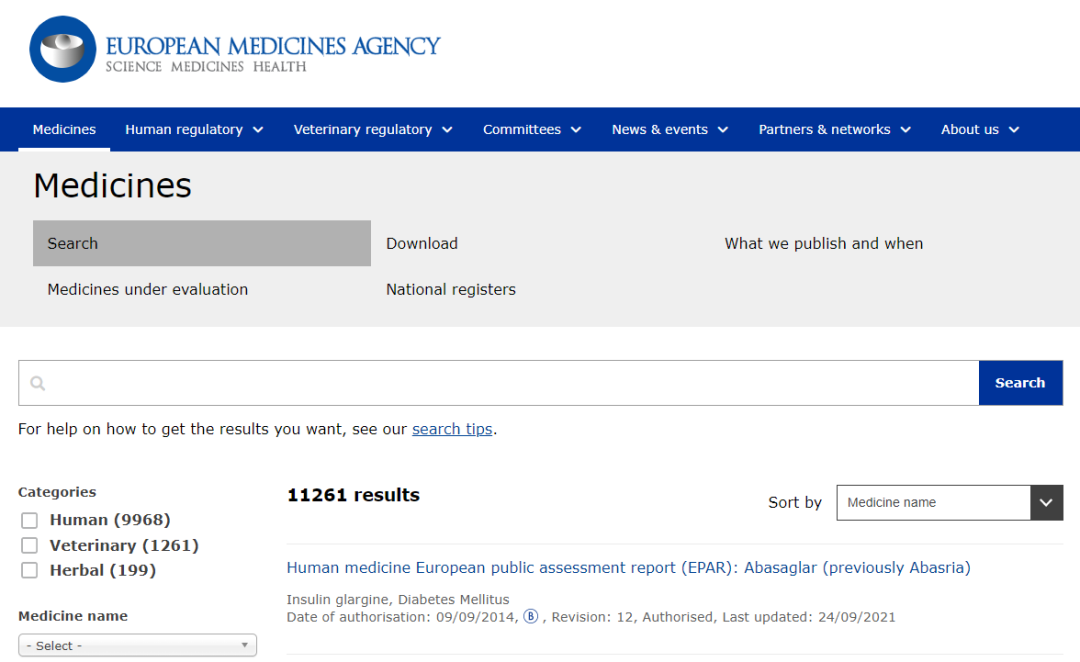
- 欧盟成员国
- Austria奥地利
http://aspregister.basg.gv.at/aspregister/faces/aspregister.jspx
- Belgium比利时
http://banquededonneesmedicaments.fagg-afmps.be/
- Bulgaria保加利亚
SmPC:
http://www.bda.bg/images/stories/documents/bdias/drugs2_list2_1.htm
Package leaflet:
http://www.bda.bg/images/stories/documents/bdias/drugs2_list2_2.htm
- Croatia克罗地亚
http://www.halmed.hr/Lijekovi/Baza-lijekova/
- Cyprus塞浦路斯
http://www.phs.moh.gov.cy/web/guest/drug-search
- Czechia捷克
http://www.sukl.eu/modules/medication/search.php?lang=2
- Denmark丹麦
http://www.produktresume.dk/AppBuilder/search
- Estonia爱沙尼亚
http://ravimiregister.ravimiamet.ee/?pv=HumRavimid.Otsing
- Finland芬兰
http://www.fimea.fi/laakehaut_ja_luettelot/laakehaku
- France法国
http://agence-prd.ansm.sante.fr/php/ecodex/index.php
- Germany德国
http://www.pharmnet-bund.de/dynamic/de/arzneimittel-informationssystem/index.html
- Greece希腊
http://www.eof.gr/web/guest/search
- Hungary匈牙利
http://www.ogyi.hu/gyogyszeradatbazis/
- Ireland爱尔兰
http://www.hpra.ie/homepage/medicines/medicines-information/find-a-medicine
- Italy意大利
https://farmaci.agenziafarmaco.gov.it/bancadatifarmaci/home
- Latvia拉脱维亚
http://www.zva.gov.lv/zvais/zalu-registrs/?&lang=en
- Lithuania立陶宛
http://vapris.vvkt.lt/vvkt-web/public/medications
- Luxembourg卢森堡
无线上资源
- Malta马耳他
http://www.medicinesauthority.gov.mt/advanced-search
- Netherlands荷兰
https://www.geneesmiddeleninformatiebank.nl/en
- Poland波兰
http://pub.rejestrymedyczne.csioz.gov.pl/
- Portugal葡萄牙
https://extranet.infarmed.pt/INFOMED-fo/index.xhtml
- Romania罗马尼亚
http://www.anm.ro/nomenclator/medicamente
- Slovakia斯洛伐克
http://www.sukl.sk/hlavna-stranka/slovenska-verzia/databazy-a-servis/vyhladavanie-liekov-zdravotnickych-pomocok-a-zmien-v-liekovej-databaze/vyhladavanie-v-databaze-registrovanych-liekov
- Slovenia斯洛文尼亚
http://www.cbz.si/cbz/bazazdr2.nsf/Search/$searchForm?SearchView
- Spain西班牙
http://cima.aemps.es/cima/publico/home.html
- Sweden瑞典
https://www.lakemedelsverket.se/sv/sok-lakemedelsfakta?activeTab=1
- 欧洲经济区成员国
- Iceland冰岛
http://www.ima.is/publications/lists/medicinal_products_with_ma_in_Iceland/
- Liechtenstein列支敦士登
参见Austria
- Norway挪威
http://www.legemiddelsok.no/
<END>


















































 川公网安备51019002008863号
川公网安备51019002008863号 本网站未发布麻醉药品、精神药品、医疗用毒性药品、放射性药品、戒毒药品和医疗机构制剂的产品信息
本网站未发布麻醉药品、精神药品、医疗用毒性药品、放射性药品、戒毒药品和医疗机构制剂的产品信息
收藏
登录后参与评论
暂无评论